Enthalpy Explained....
Enthalpy, Entropy, Pressure-Enthalpy charts, Mollier charts? I am sure there are a few of you (including myself a few years back) heard of these words, but had no real clue to their meaning, reason for understanding them, and what would we really need to understand about these strange terminology, that will allow the technician to perform his or her job any better?
I for one, in 2008, heard these terms for the very first time, and i had no clue what they meant. My problem was, and still is today, that should i come across a topic or subject, that i have very little knowledge about, i immediately get to work researching, speaking to industry related friends, engineers, just about everything to fully understand and comprehend these new found terms, and its meanings, and exactly what its reason for existence is. Most importantly, what i am missing, and how can i be the best i can be if i do not know this..... Yes, i create my own problems, i know, however it allows me to share with all of you, who hopefully will start to follow me in my blogging journey.
Lest start with Entropy...
Entropy measures the energy dispersion in a system divided by temperature. this ratio represents the tendency of energy to spread out, to diffuse, to become less concentrated in one physical location or one energetic state. That spreading out is often done by molecules because molecules above absolute zero always have energy inside of them. That's why they are incessantly speeding through space and hitting each other and rotating and vibrating in a gas or liquid. Entropy is measured in KJ/Kg/℃.
Entropy = Change in heat/temperature
= △ q / t
Energy is the capacity of a system to do work where "system" refers to any physical system, not just a refrigeration system.
Enthalpy
Enthalpy is the total amount of heat in one Kg of a substance. its units are therefore kj/Kg. The imperial part is BTU/Lb.
Mollier Charts
Mollier charts are used in designing and analyzing performance of vapour compression refrigeration systems. Each refrigerant has its own chart which is a graph of the Enthalpy of a refrigerant during various pressures and physical states. Mollier charts are also called Pressure-Enthalpy diagrams. Pressure is shown on the vertical axis, enthalpy is on the horizontal axis.
Below is an example of a Pressure - Enthalpy chart or Mollier Chart
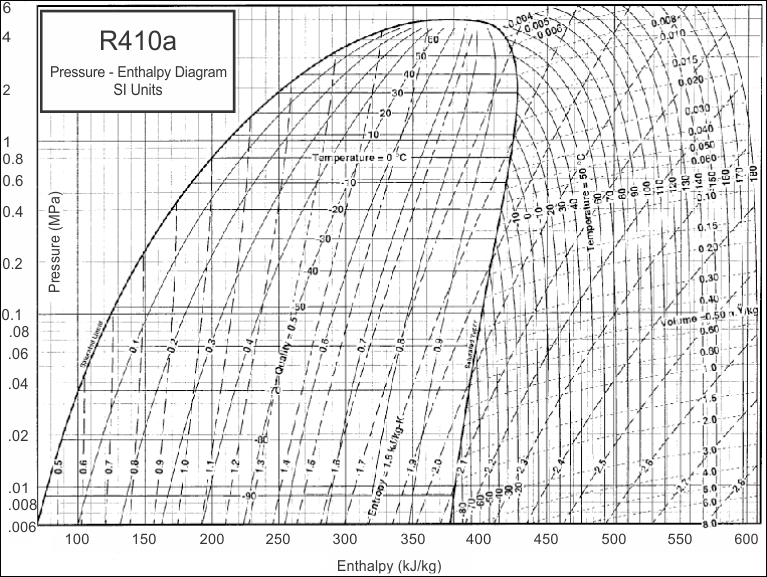
R410a Pressure - Enthalpy Chart
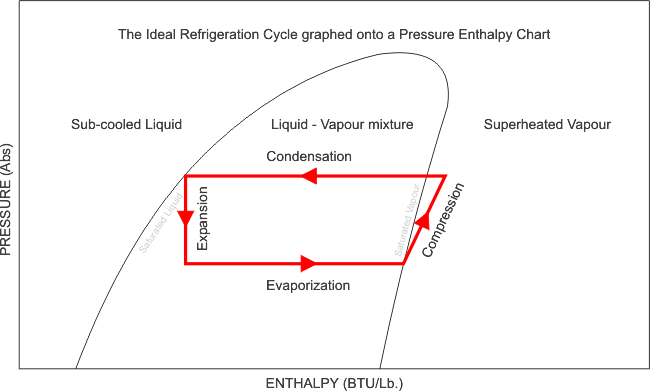



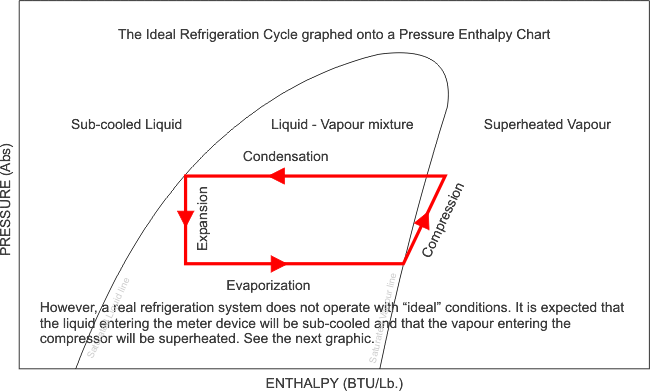
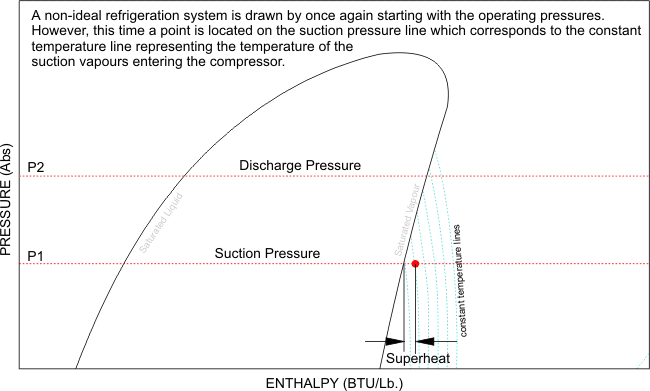



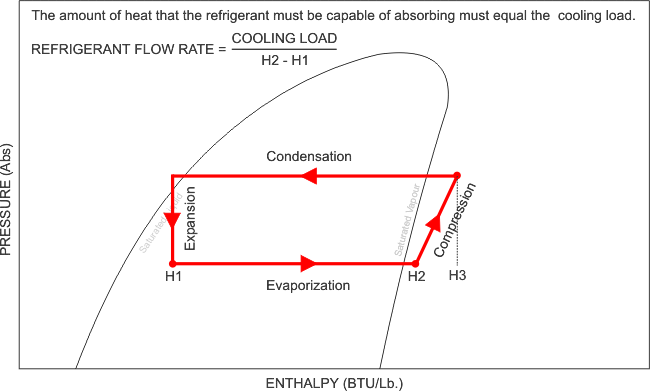
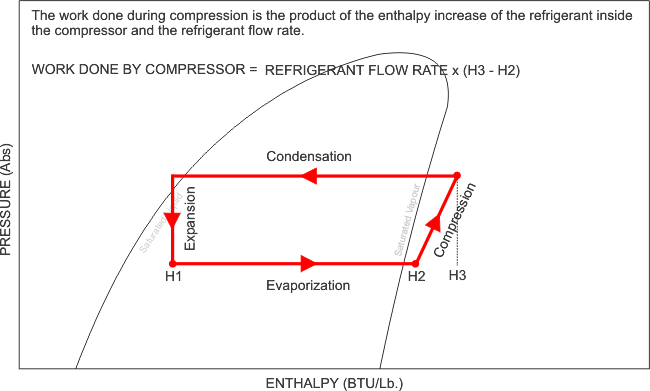


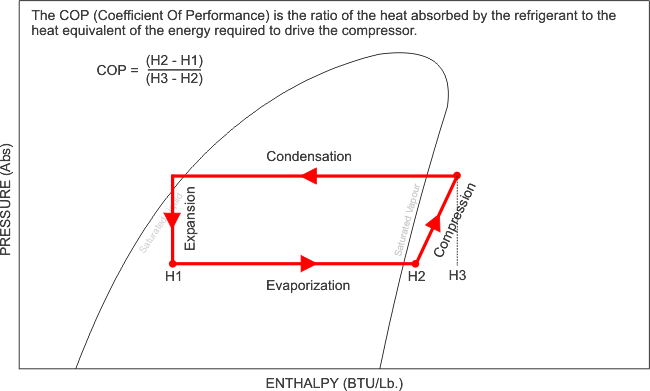
Now that i have illustrated exactly how the Pressure - Enthalpy chart looks, like, it is clear that without this understanding, we can not fully commission, re-commission a refrigeration system and be completely satisfied with its performance, after all, what are we referencing our final commissioning data to? We need the correct refrigerant chart in order to plot our recorded and measured data and perform the necessary calculations and identify whether the evaporator has the designed cooling effect, whether the heat of compression and the evaporator enthalpy equals the heat rejected by the condenser. We can also calculate the refrigerant mass flow rate, and calculate the capacity of the system.
These logs, and data should be placed in the refrigeration plant room for future reference.
I trust this blog was informative and created a new found knowledge to any one who ever questioned these terminologies. I really do hope that these blogs of mine do assist those technicians who really want to be the best they can be in this trade.
Please comment on my page www.cyrilmacquetmaurel.blogspot.com.
As usual a well researched and put together post. Great that information is shared so freely so that all an learn and become better technicians at the end of the day. Thank you Cyril
ReplyDeleteAwesome. Well done.
ReplyDeleteSo awesome that you are willing to empower others so freely.